Periphyton, Phytoplankton, and Aquatic Plants

Submerged aquatic plants at D10 ARIK. Photo credit: Stephanie Parker.
Environmental factors such as flooding, scouring, wave activity, water level, light attenuation, and nutrient availability have strong effects on the aquatic plant community. Light quantity and quality, water depth, wave activity, current velocity, and flow regime are the most important environmental factors affecting aquatic plants and bryophytes. Aquatic plants and macroalgae can alter water velocity and current, take up nutrients, settle sediments, stabilize and add complexity to the lake or stream bottom, and provide shelter and food to macroinvertebrates and fish. They can also act as indicators of changes in watershed activity by integrating the effects of changing nutrient loads, toxicity, and land cover.
As a key part of the aquatic ecosystem, it is important to sample these primary producers to determine changes in community structure, invasive species extent, abundance, and biodiversity over time, as well as changes in biogeochemical cycles. Data on periphyton (organisms that live attached to underwater surfaces) and phytoplankton (organisms that live in the water, unattached to surfaces) can be used to track algal abundance, density, and biomass, as well as community structure and function and the introduction of invasive species.
NEON samples primary producers, or organisms that convert energy from an abiotic source, such as the sun, in the aquatic algal and plant communities. Aquatic plants, bryophytes, lichens, and macroalgae, along with algae and microbes, form the base of the food web. Presence/absence, composition, and abundance data for aquatic plants, bryophytes, lichen, and macroalgae are also collected at aquatic field sites throughout the year. In addition, NEON conducts biogeochemical analyses and archives a subset of specimen samples at the NEON Biorepository. These data are compatible with data from existing environmental monitoring programs and will provide a long time-series of seasonal data across NEON aquatic sites.

Domain 13 field ecologist scrubs periphyton on a cobble at the COMO field site (photo by Stephanie Parker)
Sampling Design
Sampling Collection
NEON collects algae and aquatic plant data at all aquatic sites. The goals of periphyton, phytoplankton, and aquatic plant sampling are to determine species diversity, taxa richness, and biomass. Plant point count data can also be used to determine percent cover of plants across aquatic sites.
Periphyton and phytoplankton
Sampling is conducted using a percent-based habitat approach, where samples and data are collected in two different habitat types. Wadeable stream sites are divided into habitats such as riffles, runs, pools, and step pools, while samples from lakes and rivers are collected in littoral (nearshore) areas and in the water column. All benthic (stream, lake bottom) samples are collected from the surface of the natural substratum present (e.g., cobbles, sand, silt) and phytoplankton samples are collected from the water column in lakes and rivers.
Benthic field collection methods differ depending on the substrata being sampled. Field protocols and sampling methods differ depending on the habitat and substrate being sampled. All samples are collected from the stream, river, or lake bottom or water column in each habitat using standard quantitative and semi-quantitative sampling methods. Field samples are subsampled and processed in the Domain support facility within 24 hours of collection and sent to taxonomy and chemistry labs.

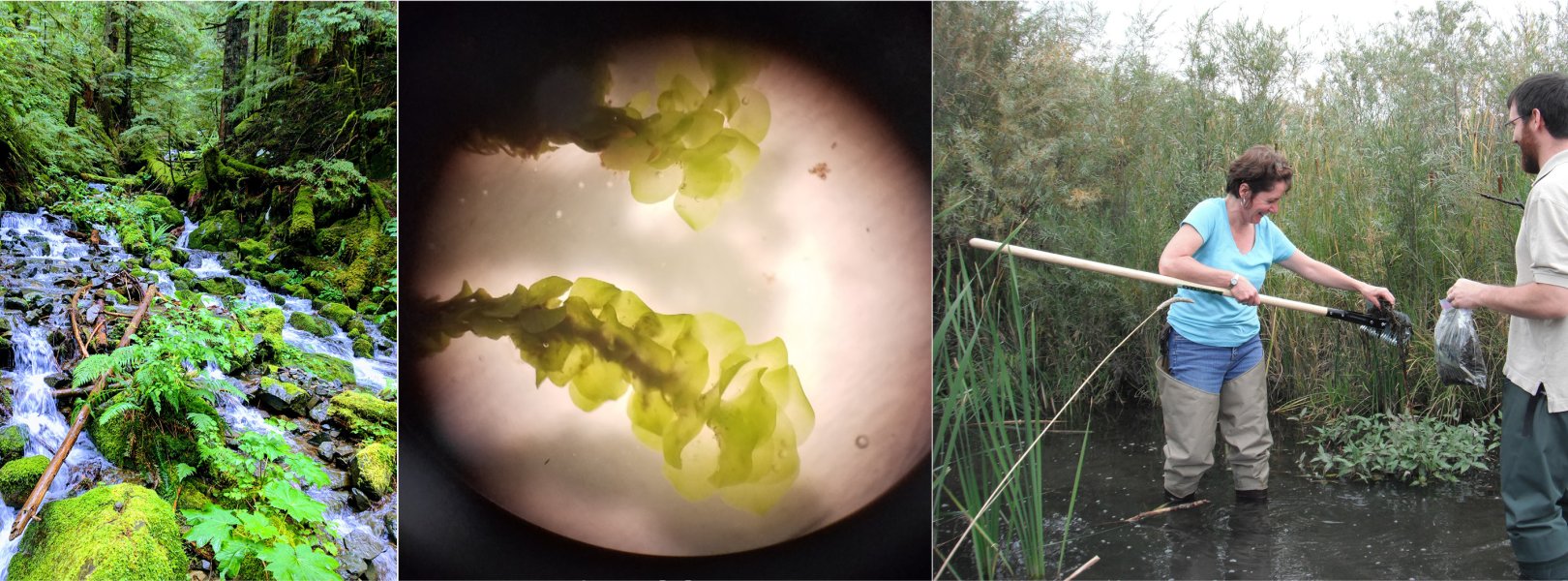
Domain 05 field ecologist prepares to collect aquatic plants at the CRAM lake field site using a rake. Photo credit: Hannah Beeler.
Aquatic plants
Aquatic plant sampling in wadeable streams is conducted at ten permanent transects placed during the first year of sampling at a site. Transects are located in two different habitat types at the time of placement. However, those habitats may change over time as water levels fluctuate. Plant sampling locations in lake and river sites are randomized and placed in areas at depths where plants are known to grow. Because water and objects in the water (including particulates) filter out light, plants cannot grow below certain depths depending on the body of water. Plants are identified by NEON field ecologists where possible. When specimens cannot be identified with certainty, they are sent to an expert taxonomist for verification.
Data Products
The periphyton and phytoplankton collection data products includes information on subsampling and quality control, taxonomic identification, abundance, and biomass. The collection field data are also contain parent records for algal biogeochemical data.
- Periphyton, seston, and phytoplankton collection (DP1.20166.001)
- Periphyton, seston, and phytoplankton chemical properties (DP1.20163.001)
Plant point count data include information on wadeable stream, transect-based point counts, including abundance and species identification for vascular and nonvascular plants as well as macroalgae.
The aquatic plant clip harvest data include field metadata for biomass, species identification, and biogeochemical data products. Clip harvest and biomass data are produced during mid-summer sampling at all sites; however, lake and river presence/absence data are available in this data product for the spring and fall sampling bouts as well.
- Aquatic plant bryophyte macroalgae clip harvest (DP1.20066.001)
- Aquatic plant bryophyte chemical properties (DP1.20063.001)
Archival Samples
Archival samples of periphyton, phytoplankton, macroalgae, and aquatic plants collected at NEON aquatic field sites are available from the NEON Biorepository.
| Types of Samples | Storage Conditions | Storage Container | Quantity Archived (Annually) † | Link to the Collection |
|---|---|---|---|---|
| Algae: Periphyton, seston, phytoplankton – soft algae | glutaraldehyde | 60 mL jar or smaller |
600 to 800 samples |
Aquatic Microalgae Collection (Chemical Preservation) (NEON-AMIC-CP) |
| Algae: Periphyton, seston, phytoplankton – diatoms | freeze dried | vials | 600 to 800 samples |
Aquatic Microalgae Collection (Freeze-dried) (NEON-AMIC-FD) |
| Algae: Periphyton, seston, phytoplankton – diatoms | dry | slides | 600 to 800 slides |
Aquatic Microalgae Collection (Microscope Slides) (NEON-AMIC-MS) |
| Aquatic macrophyte vouchers | dry | herbarium paper | 70 to 370 specimens |
Aquatic Plant, Bryophyte, and Lichen Collection (Herbarium Vouchers [Standard Sampling]) (NEON-APLC-HVSS) |
| Aquatic mosses/lichens/ liverworts | dry | herbarium packet | included above | included above |
| Macroalgae | glutaraldehyde | 60 mL jar | 150 to 300 specimens |
Aquatic Macroalgae Collection (Chemical Preservation [Point Counts]) (NEON-AMAC-CPPC) |
† Quantities represented in this table are annual quantities expected from sampling conducted throughout the observatory during full Operations. Actual quantities may differ from these projections